MANCO Project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 101003651
MANCO RESEARCH

Scientific objectives
THE SPECIFIC OBJECTIVES OF THE MANCO PROJECT
ARE AS FOLLOWS:
1
To screen in vitro and identify H2L2 monoclonal antibodies against SARS-COV-2 with potential prophylactic and/or therapeutic properties, suitable for rapid manufacturing based on the IMI-ZAPI methodology pipeline.
2
To identify relevant animal models for the in-vivo evaluation of (prophylactic and/or therapeutic) H2L2 monoclonal antibodies against SARS-COV-2 challenge and for safety evaluation, including absence of ADE (Adverse Drug Effect).
3
To implement rapid manufacturing lines of production using the C1 fungal production platform developed by Dyadic expressing high yield antibody.
4
To ensure regulatory adequacy for emergency use during outbreaks.
5
To advance lead product(s) in order to reach preclinical package completion.
Overall structure of work plan
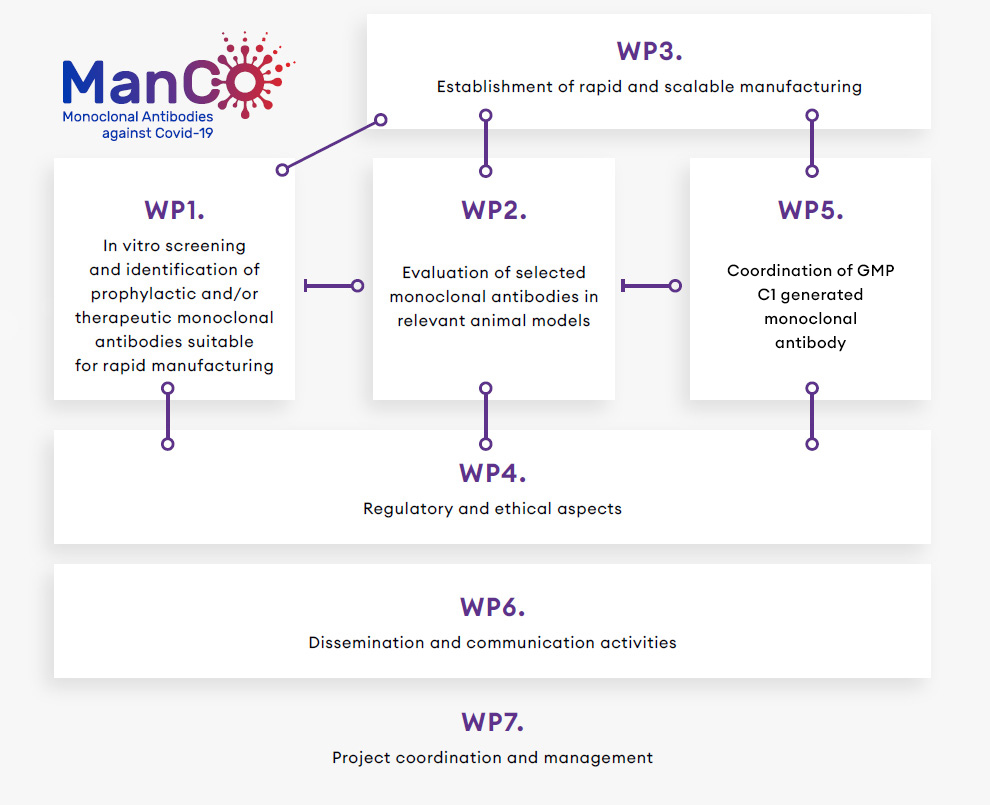
The overall structure of the MANCO project work plan relies on the establishment of 7 distinct yet inter-connected work-packages (WPs) each with defined objectives and associated tasks. Each WP has a leading organisation which is responsible for its overall progress and reporting.

WP1.
Leading organisation: UU; WP Participants: EMC, Harbour
WP1 aims at the identification and in vitro characterization of neutralizing monoclonal antibodies against SARS-COV-2.
WP2.
Leading organisation: EMC; WP Participants: Harbour, Tiho, CSIC
WP2 evaluates the preclinical efficacy, safety and absence of ADE in relevant animal models.
WP3.
Leading organisation: Harbour; WP Participant: CR2O
WP3 implements rapid and scalable manufacturing lines for the production of research-grade, GLP-grade and GMP (clinical grade) batches of the selected monoclonal antibodies to enter pre-clinical evaluation in WP2, GLP compliant toxicity studies, and Phase I clinical study.
WP4.
Leading organisation: IABS-EU
WP4 ensures regulatory adequacy of the proposed approach for eventual emergency use.
WP5.
Leading organisation: CR2O
WP5 coordinates GMP C1 generated monoclonal antibody.
WP6.
Leading organisation: EMC; WP Participant: FINOVATIS
WP6 coordinates MANCO’s dissemination and communication strategy.
WP7.
Leading organisation: EMC; WP Participant: FINOVATIS
WP7 is dedicated to the project’s coordination and management.
 MANCO Project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 101003651
MANCO Project has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 101003651